Management of Oral Secretions and Congestion at End of Life
This document is for informational and educational purposes only and is not a substitute for medical advice, diagnosis, or treatment provided by a qualified health care provider. All information contained in this document is protected by copyright and remains the property of ProCare HospiceCare. All rights reserved.
The presence of noisy respirations due to excessive secretions (also known as “gurgling” or “rattling”) at end of life predict a prognosis of days to weeks and can be alarming to family members.

The presence of noisy respirations due to excessive secretions (also known as “gurgling” or “rattling”) at end of life predict a prognosis of days to weeks and can be alarming to family members. Typically, patients do not require treatment for terminal secretions because it is not bothersome to them. However, the noises are bothersome to the caregivers, and their number one goal is ensure the comfort of their loved one in their last days.
Mucous is composed of 95% water, glycoproteins, proteoglycans, and lipids. In normal circumstances, we produce about 1.5 quarts of mucous per day, pushed to the back of the throat by tiny cilia and swallowed (this will occur approximately 20 times while you read this article). Mucous has two functions: it keeps the nasal cavity and air entering the lungs moist to prevent dryness, and it acts as a filter for your lungs. A healthy patient is able to effectively clear these oral secretions. However, as a patient declines, they lose the ability to swallow effectively, often due to loss of consciousness. The secretions then pool in the throat and the oscillation (movement) of air causes the noise.
There are two secretory subtypes, Salivary and Bronchial.

Certain contributing factors can increase or exacerbate secretions/congestion when a patient is admitted to hospice. These include: neurological disorders; use of certain medications (clozapine, clobazam, and/or doxycycline); cancer of the mouth, tongue, larynx, or esophagus; respiratory edema or infection; and also pre-existing gastroesophageal issues such as dysmotility or gastroesophageal reflux disorder (GERD).
The best way to tackle end-stage secretions is to follow these three steps:
- Determine the type of the secretion
- Start with non-pharmacological options first
- Add on a preferred medication based on patient-specific factors
Non-Pharmacological Interventions
- Re-positioning the patient on their side can facilitate drainage
- Reducing fluid intake or intravenous fluid administration
- Maintaining adequate oral hygiene
- Gentle suctioning may be used; this may be ineffective when fluids are beyond reach, and can be disturbing to both the patient and surrounding caregivers
- Communication with family and caregivers to explain terminal secretions
Pharmacological Interventions
- Drug selection should be individualized
- Selecting which medication to use should be based on patient factors (such as age, diagnosis, prognosis) as well as drug factors (onset of action, route of administration, cost)
- Manage symptoms of underlying pathology (e.g. CHF, lung disease)
- Consider an antibiotic, if appropriate, based on prognosis, allergies, patient/family goals of care
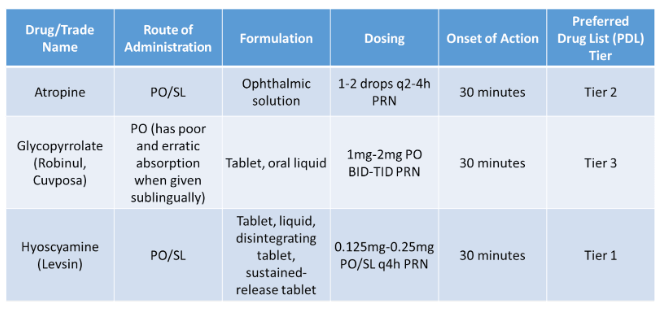
Oral/Sublingual Treatment Options
Treatment Options Using Alternative Routes
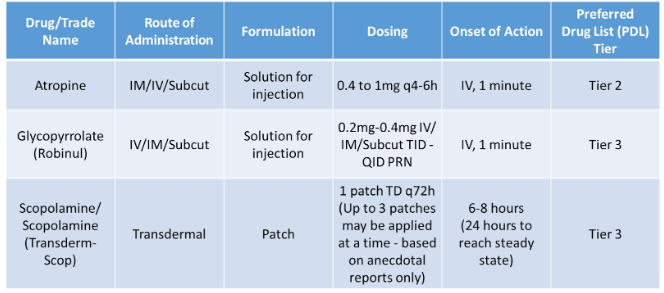
Utilize anticholinergic medications with caution, as the risk of side effects may outweigh the benefits of their use to lessen secretions. Common anticholinergic side effects include: blurry vision, dry mouth, confusion, tachycardia, urinary retention, skin flushing and constipation. Atropine and scopolamine are both tertiary amines and readily cross the blood-brain barrier (BBB), which may lead to increased CNS side effects such as confusion, delirium, hallucinations, sedation, and agitation. Hyoscyamine and glycopyrrolate are both quaternary amines and do not cross BBB and, therefore, less likely to cause CNS side effects.
Scopolamine is available as a patch and has issues with unpredictable absorption, as well as difficulties with titration. It can take up to 6-12 hours for the desired effects and, in some studies, up to three patches may be applied to aid in secretion management.
Hyoscyamine is the anticholinergic of choice, 0.125mg SL Q4H prn secretions, due to lower cost and availability as a sublingual tablet. Recommend monitoring patients for adverse effects and avoid use with bronchial secretions, as use can create a mucous plug and worsen secretions.
When chest congestion is suspected vs. end-stage secretions, guaifenesin (Mucinex) may be utilized as an expectorant to help loosen and expel thicker secretions. Typical dosing is 600-1200mg PO BID for the extended release tablet, and 20mL PO Q4H prn for congestion for the liquid. Keep in mind that guaifenesin should be taken with plenty of water and can cause nausea as a side effect.
Alternative Treatments for Chest Congestion:
- Nebulized solutions:
- Bronchodilator: Albuterol 0.083% 1 unit dose INH Q4H prn congestion
- Hypertonic saline 3% via nebulizer (use albuterol first, then saline)
- Alternative Bronchodilator: Theophylline (use with caution due to adverse reactions)
- Antibiotics: If indicated and in line with goals of care (limited due to culture/sensitivity results)
- N-acetylcysteine (Mucomyst): Reserve for most refractory cases
- $$$, odorous, risk of bronchospasm (use with bronchodilator, if possible)
End-stage secretions are commonly seen in hospice and palliative care patients, and assessment is key to successfully treating this bothersome symptom. It is important to determine if they are truly respiratory secretions or congestion related to an underlying respiratory issue, such as infection or a COPD exacerbation. There are several options available to treat secretions and, as always, it is important to consider risk vs. benefit, especially prior to starting an anticholinergic medication with a well-known side effect profile. Monitor the patient closely and be proactive with medications on hand in the care pack or on standing orders to help keep the patient comfortable 24/7/365.
References:
- Hsin G and Hallenbeck J. Fast Facts and Concepts #158. Respiratory Secretion Management. Palliative Care Network of Wisconsin. [Online] Available from: https://www.mypcnow.org/fast-fact/respiratory-secretion-management.
- Bickel K, Kareem L et. Al. Fast Facts and Concepts #109. Death Rattle and Oral Secretions. Palliative Care Network of Wisconsin. [Online] Available from: https://www.mypcnow.org/fast-fact/death-rattle-and-oral-secretions/.
- Kintzel PE, Chase SL et. Al. Anticholinergic medications for managing noisy respirations in adult hospice patients. Am J Health-Syst Pharm. 2009; 66:458-64.
- Morrison L, Innes S. Oscillating devices for airway clearance in people with cystic fibrosis. Cochrane Database Syst Rev. 2017 May 4;5(5):CD006842. doi: 10.1002/14651858.CD006842.pub4. Update in: Cochrane Database Syst Rev. 2020 Apr 30;4:CD006842. PMID: 28471492; PMCID: PMC6481377.
- Back IN, Jenkins K, Blower A, Beckhelling J. A study comparing hyoscine hydrobromide and glycopyrrolate in the treatment of death rattle. Palliat Med 2001; 15: 329-336.
- Bennett M, Lucas V, Brennan M, et al. Using anti-muscarinic drugs in the management of death rattle; evidence based guidelines for palliative care. Palliat Med 2002; 16:369-374.










